Update Tavneos Review – April 2026
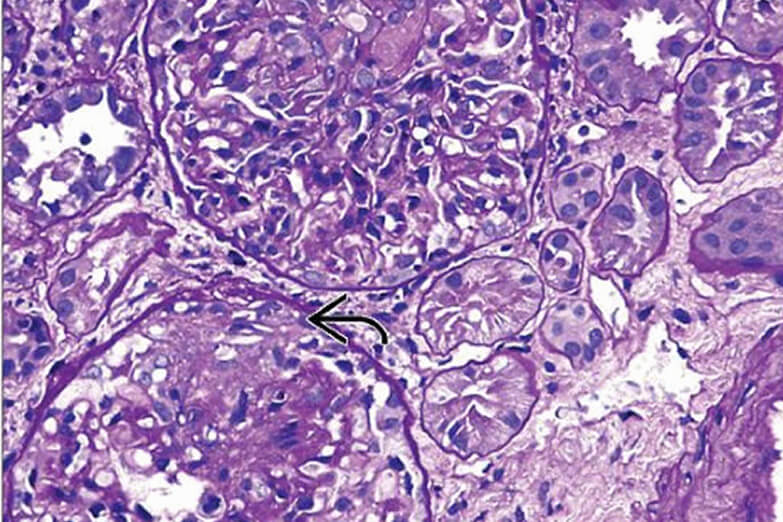
The U.S. Food and Drug Administration (FDA) issued a Drug Safety
Communication on 31 March 2026, alerting healthcare professionals and patients to serious cases of drug-induced liver injury (DILI) in postmarketing surveillance of avacopan — including cases with fatal outcomes.
While the FDA operates outside the European regulatory framework,
its postmarketing findings are directly relevant to the EMA’s ongoing assessment.